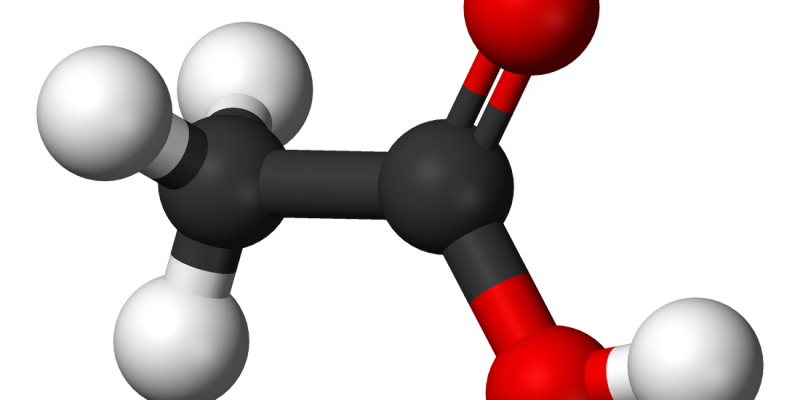
Acetic acid is one of the simplest carboxylic acids with the chemical formula CH3COOH. The systematic IUPAC name of this organic compound is ethanoic acid. It forms ice-like crystals temperatures below 16.6oC. Acetic acid is made up of two functional groups – a methyl group, and a carboxyl group. Some important properties and uses of acetic acid are discussed in this article.
Properties of Acetic Acid
The molar mass of acetic acid is 60.052 grams per mole. It has a colorless appearance and is liquid at standard conditions for temperature and pressure. It has a pungent odor which is similar to the smell of vinegar. The density of acetic acid in its liquid state is 1.049 grams per cubic centimeter. In its solid state, the density of CH3COOH increases to 1.27 grams per cubic centimeter.
The melting point of this organic compound corresponds to 17oC whereas its boiling point approximates to 119oC. It forms a homogeneous solution when dissolved in water. This compound can dissociate into a proton and an acetate ion (CH3COO–). However, it is said to be a weak acid since the extent of dissociation is not very high. For example, a solution of acetic acid in water with a molarity of 1.0M has a pH of 2.4 (click here to learn about the relation between normality and molarity)
Uses of Acetic Acid
Vinegar is essentially a solution of acetic acid and water. Acetic acid is also used in the production of the vinyl acetate monomer. Esters of this compound can be used as solvents for several coatings and paints. Glacial acetic acid serves as an excellent polar and protic solvent. The injection of acetic acid into tumors is a method of cancer treatment. To learn more about this organic compound and other important concepts in organic chemistry, subscribe to the BYJU’S YouTube channel and enable notifications.